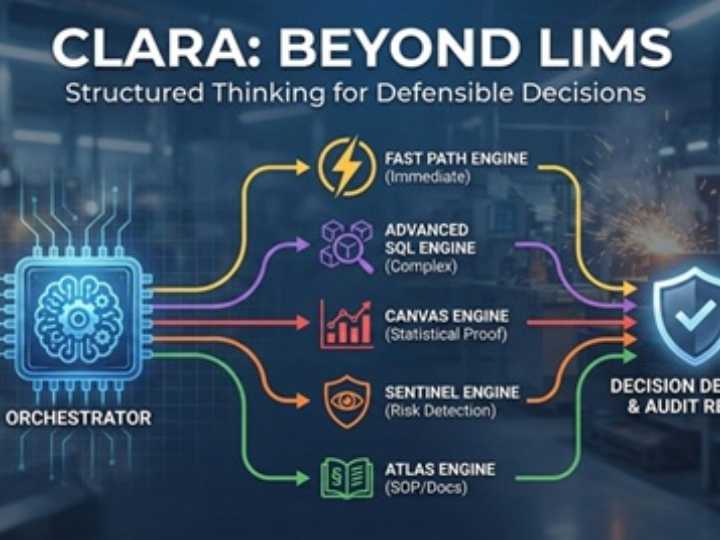
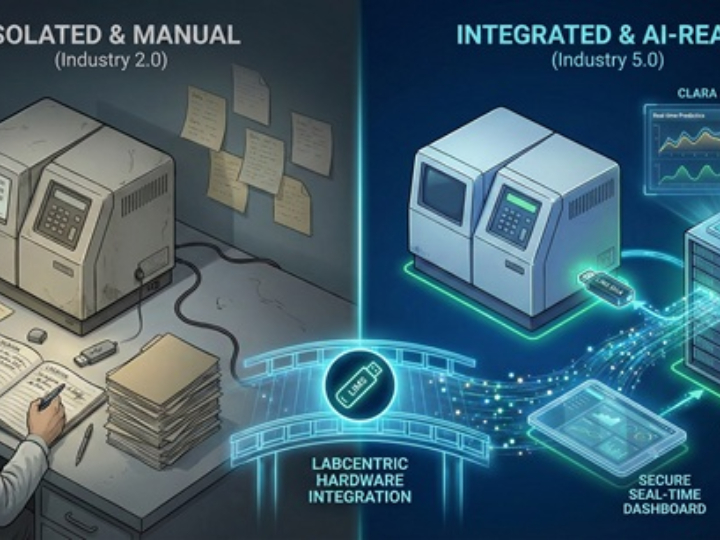
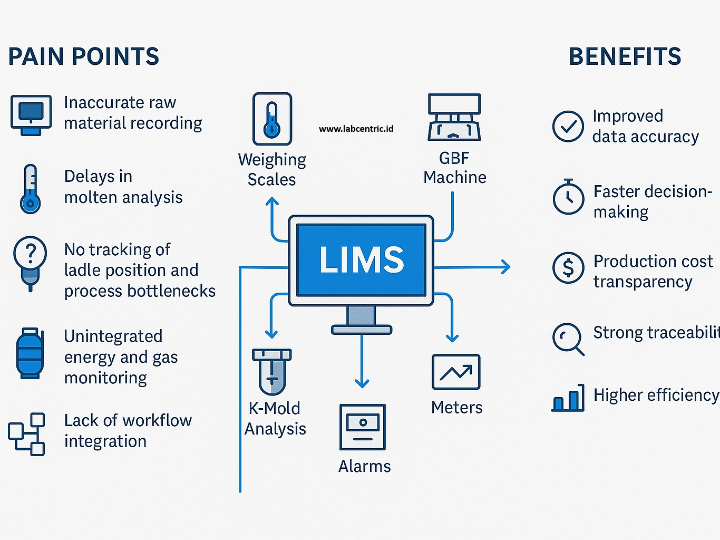
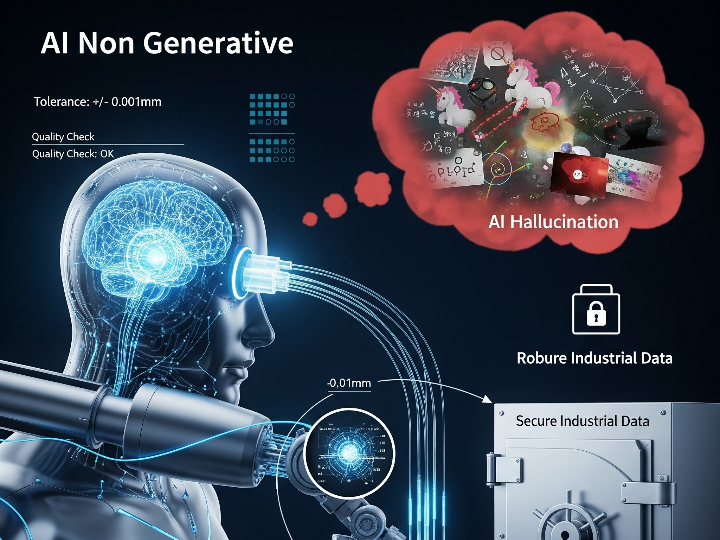
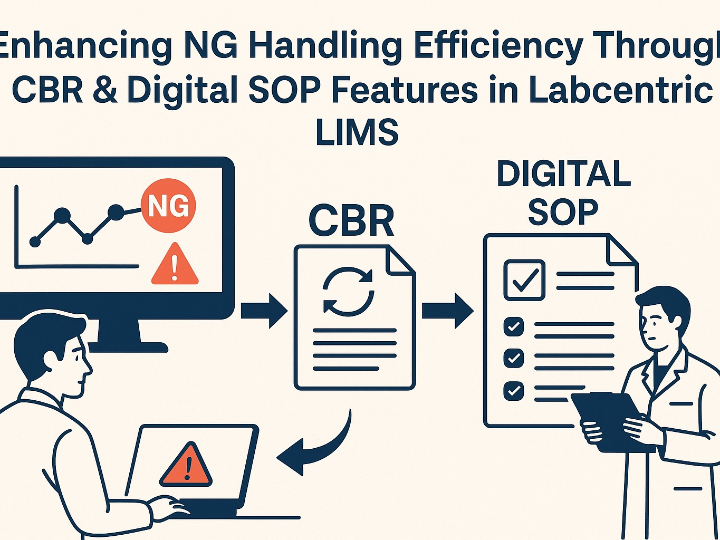
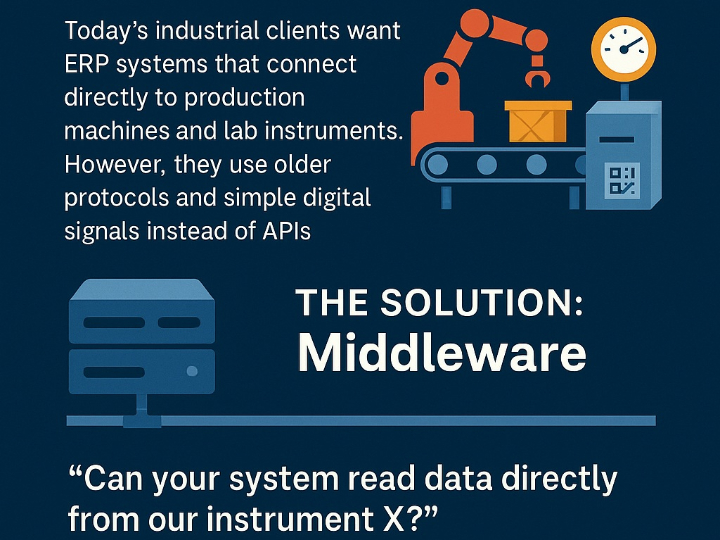
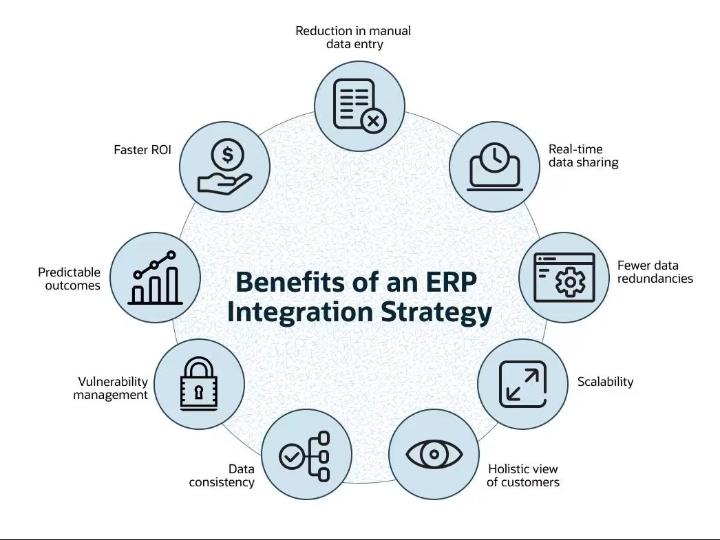
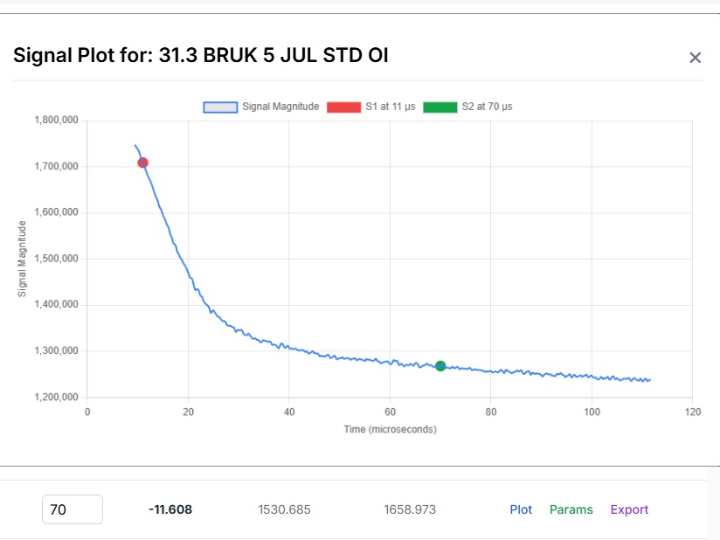
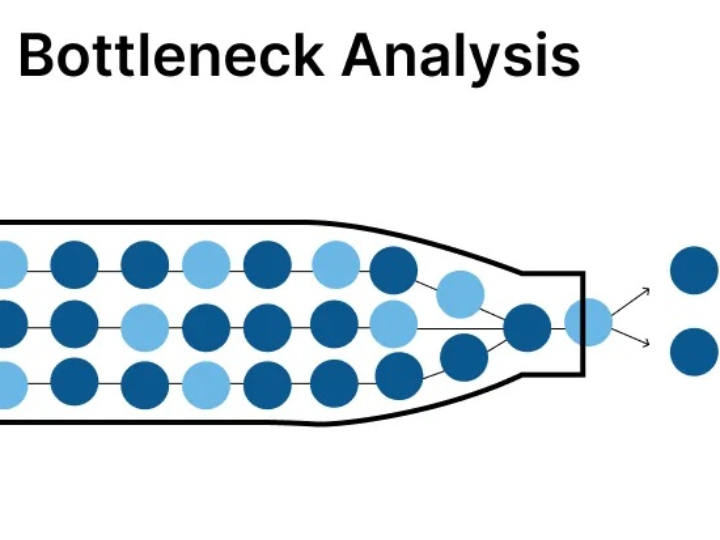
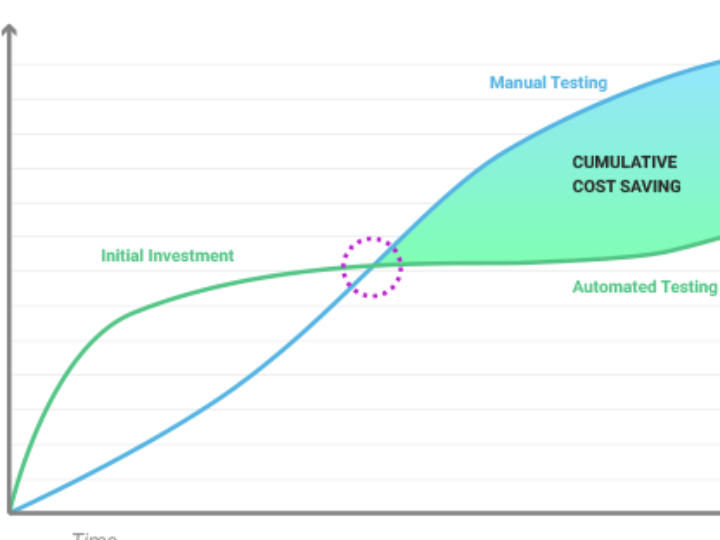
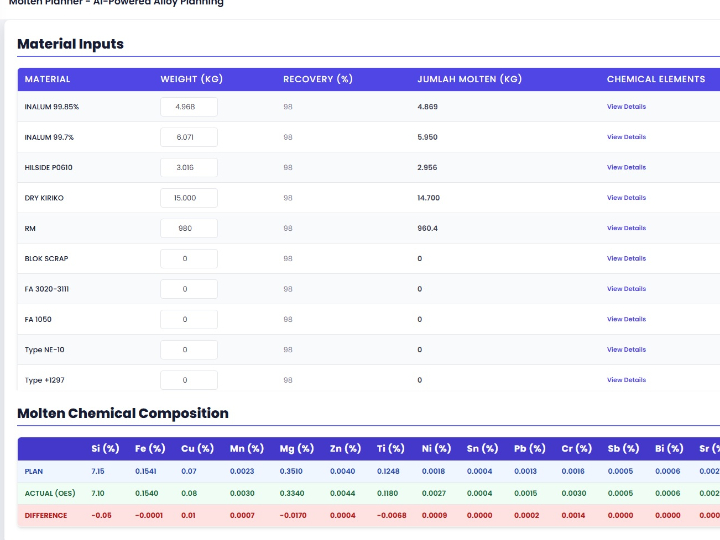
CLARA: The Future of AI-Powered Industrial Laboratory Intelligence
CLARA is a state-of-the-art AI ecosystem designed to transform industrial laboratory operations into a data-driven, autonomous environment. This guide outlines the core analytical engines that empower teams to achieve excellence in quality, safety, and efficiency.